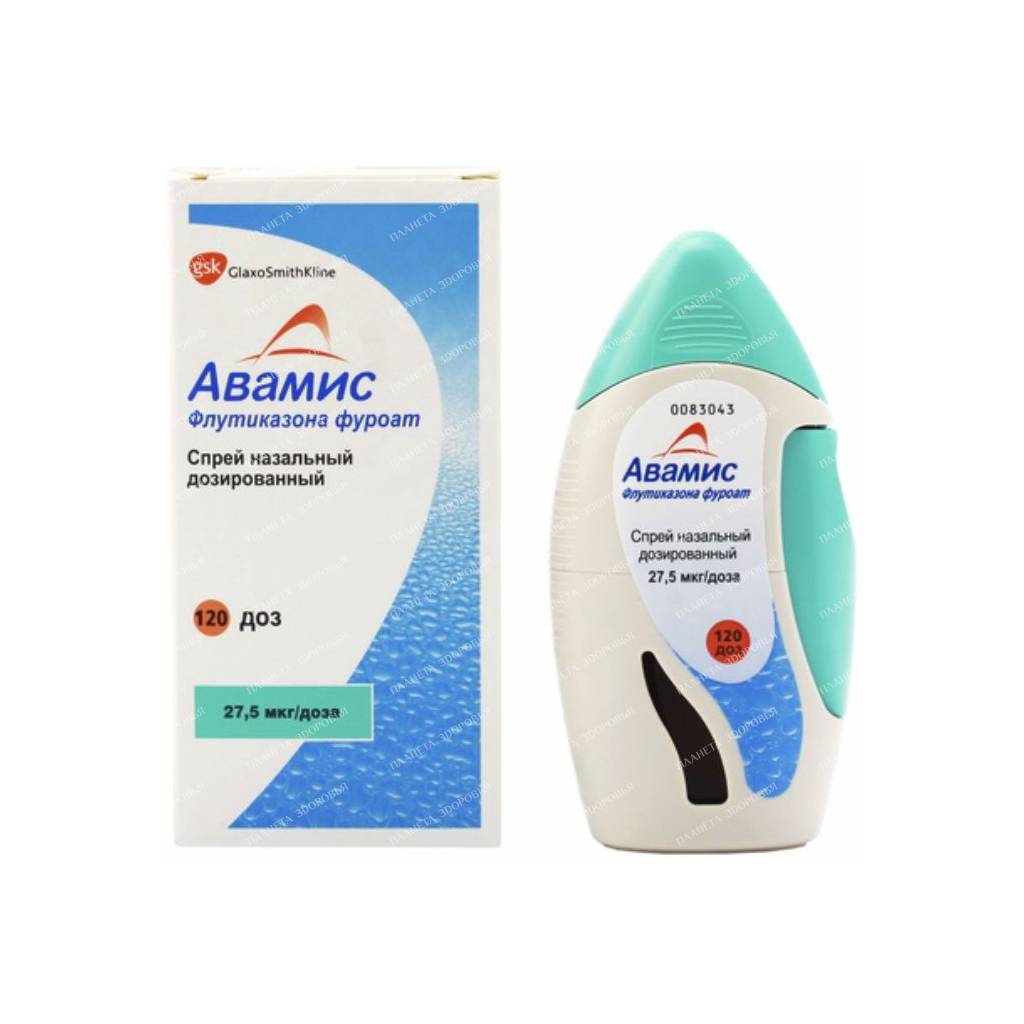
Avamys Nasal Spray 27.5 mcg/dose: Relieve Your Nasal Allergies
Get Effective Relief from Allergic Rhinitis with Avamys Nasal Spray.
Description:
Avamys nasal spray, dosed at 27.5 mcg/spray, is a potent anti-inflammatory medication that provides long-lasting relief from the symptoms of allergic rhinitis. It contains fluticasone furoate, a synthetic corticosteroid that works by reducing inflammation in the nasal passages.
Key Features:
- Fast and Effective Relief: Experience noticeable relief from your allergy symptoms, such as sneezing, runny nose, itchy eyes, and nasal congestion, within just 8 hours of the first spray.
- Convenient and Easy to Use: Avamys is a nasal spray that is simple to administer and can be used once daily for optimal results.
- Proven Efficacy: Clinical studies have demonstrated the effectiveness of Avamys in effectively controlling the symptoms of seasonal and perennial allergic rhinitis in adults, adolescents, and children aged 6 years and older.
How Avamys Works:
Fluticasone furoate, the active ingredient in Avamys, binds to glucocorticoid receptors, leading to a powerful anti-inflammatory effect. This reduces the release of inflammatory chemicals that cause allergy symptoms, providing relief from congestion, itching, and other nasal irritations.
Dosage:
- Adults and Adolescents (12 years and older): Start with 2 sprays (27.5 mcg per spray) in each nostril once a day (110 mcg total). Once symptoms are under control, the dose may be reduced to 1 spray in each nostril once a day (55 mcg total).
- Children aged 6 to 11 years: Start with 1 spray in each nostril once a day (55 mcg total). If needed, the dose may be increased to 2 sprays in each nostril once a day (110 mcg total).
Safety and Side Effects:
Avamys is generally well tolerated. The most common side effects are mild and include nosebleeds, nasal irritation, and headaches. Serious side effects are rare.
Consult your doctor or pharmacist if you have any concerns or experience any unusual side effects.
Important Information:
- Not for children under 6 years old.
- Use with caution in pregnant and breastfeeding women.
- Consult your doctor before using Avamys if you have any pre-existing medical conditions.
Get the relief you need from your nasal allergies with Avamys nasal spray. Ask your doctor if Avamys is right for you.