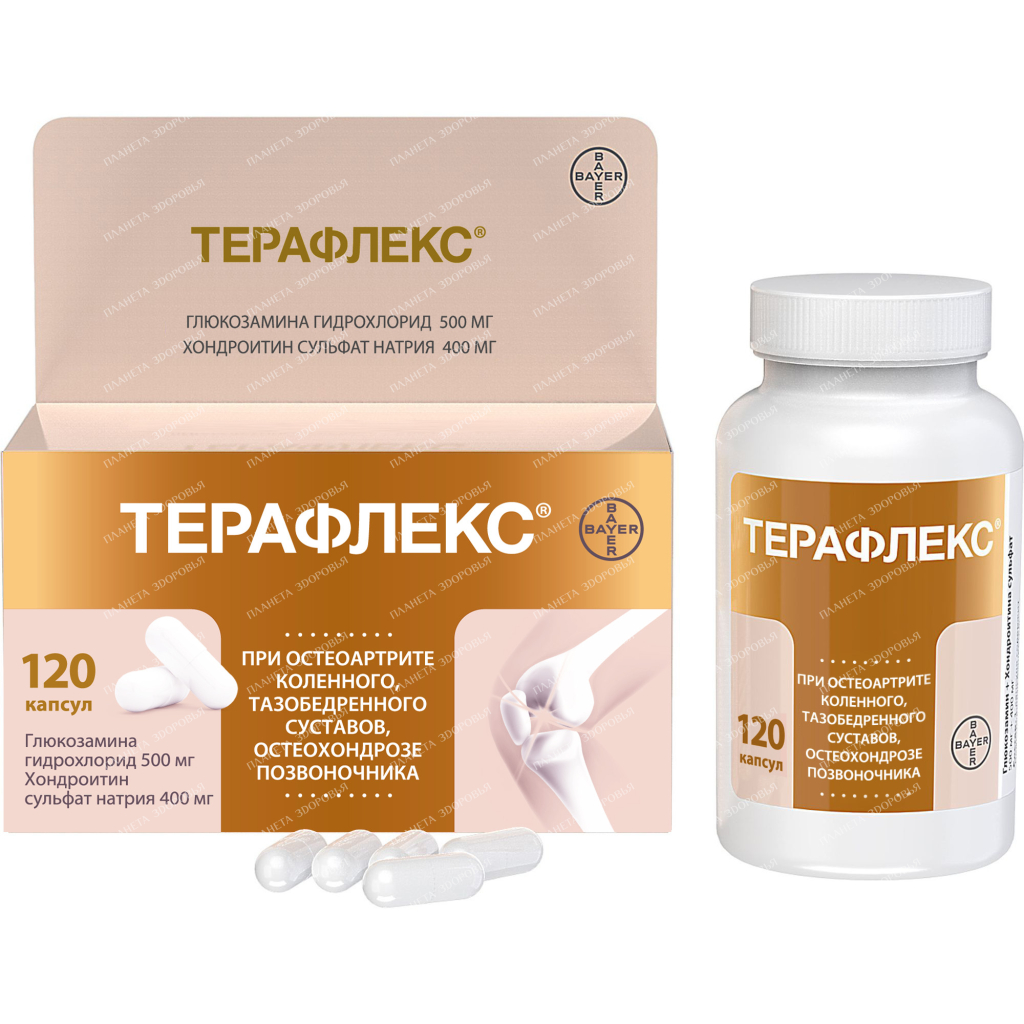
Teraflex Capsules: Powerful Joint Support for Pain Relief (500mg/400mg, 120 Capsules)
Experience relief from osteoarthritis pain with Teraflex capsules, a powerful combination of glucosamine and chondroitin sulfate. This trusted formula helps support healthy joint function and reduce discomfort for a better quality of life.
Key Benefits:
- Effective Pain Relief: Helps reduce mild to moderate osteoarthritis pain in the knees, hips, and spine.
- Supports Joint Health: Provides essential building blocks for cartilage, helping maintain its structure and function.
- Convenient Dosage: Easy-to-swallow capsules for convenient daily use.
- Clinically Studied: Backed by scientific research demonstrating its effectiveness.
What is Teraflex?
Teraflex capsules contain two key ingredients:
- Glucosamine: A naturally occurring substance that helps build cartilage and reduce inflammation.
- Chondroitin Sulfate: A building block of cartilage that helps absorb shock and lubricate joints.
Who is Teraflex for?
Teraflex is recommended for adults experiencing discomfort from osteoarthritis. It is not recommended for children under 18 years of age.
How to Use Teraflex:
The recommended dosage is 1 capsule 3 times a day, providing 1000-1200 mg of chondroitin sulfate and 1500 mg of glucosamine. Treatment should be continued for at least 3-6 months for optimal results.
Important Information:
- Consult your doctor before using Teraflex, especially if you have existing medical conditions.
- Teraflex may interact with certain medications, so inform your doctor about all medications you are taking.
- If you experience any side effects, stop using Teraflex and consult your doctor immediately.
Start feeling the difference today! Order your Teraflex capsules now.
Buy Teraflex capsules 500mg/400mg №120 №1
| INN | CHONDROITIN SULPHATE+GLUCOSAMINE |
|---|---|
| The code | 50 362 |
| Barcode | 4 250 369 503 803 |
| Dosage | 500mg/400mg |
| Active substance | Chondroitin sulfate, glucosamine |
| Manufacturer | Bayer Consumer Care AG, Switzerland/ Contract Pharmacal Corp., USA |
| Trademark | Bayer |
| trade line | Teraflex |
| Importer | Foreign trade unitary enterprise "BELTRANSFER-MED", 220073 Minsk, Olshevsky str., 20/11-24, 8th floor, room 20 |
Related products
Specialty Supplements
Specialty Supplements
Specialty Supplements
Specialty Supplements
Specialty Supplements
 Free worldwide shipping on orders $99+
Free worldwide shipping on orders $99+  US: temporary delays — postal services aligning new import rules,
US: temporary delays — postal services aligning new import rules,  EU: 1–2 weeks,
EU: 1–2 weeks,  Worldwide: 1–4 weeks
Worldwide: 1–4 weeks