Subtotal: $161.50
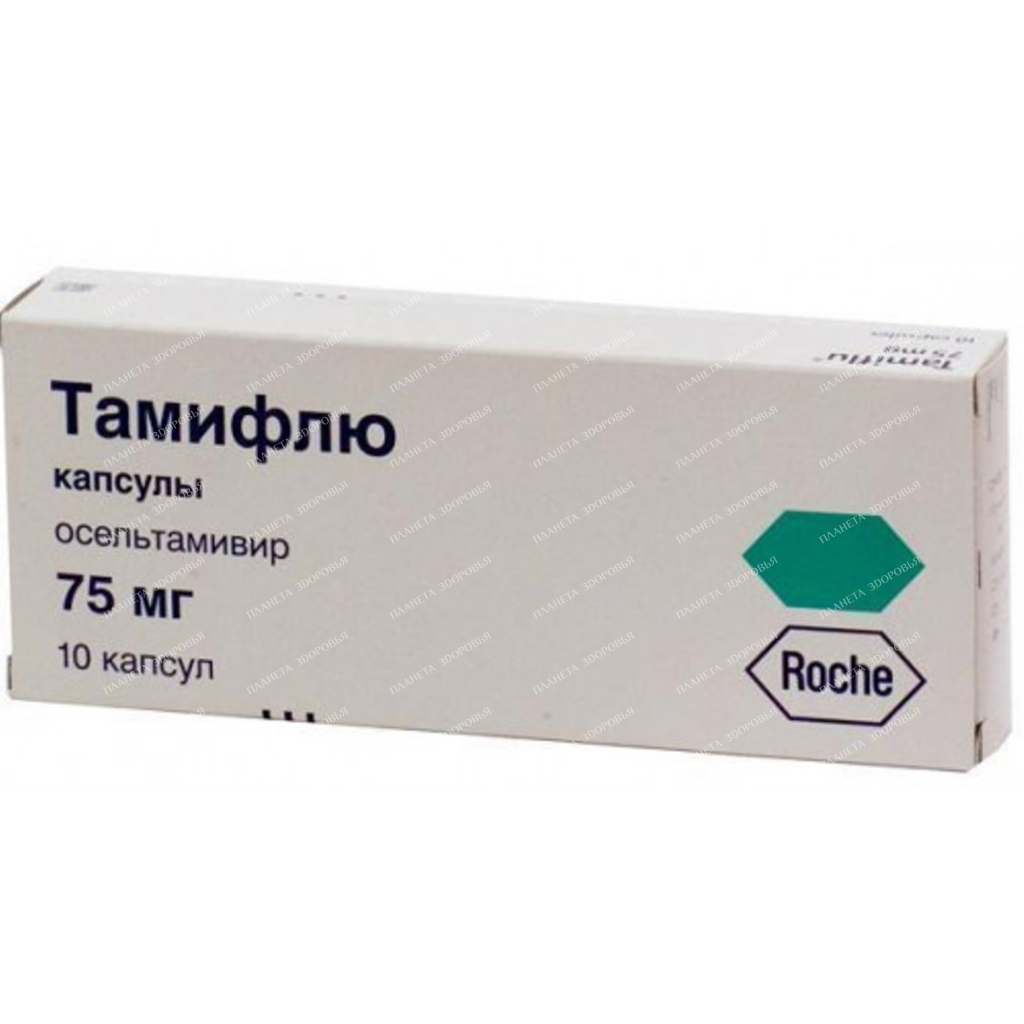
Tamiflu Capsules 75mg: Effective Influenza Treatment & Prevention
Tamiflu 75mg capsules are a potent antiviral medication used to treat and prevent influenza A and B infections in adults and children over 1 year old. This comprehensive guide provides detailed information on its usage, dosage, side effects, and interactions.
What is Tamiflu?
Tamiflu contains oseltamivir phosphate, a prodrug that converts to the active metabolite oseltamivir carboxylate (OK). OK is a selective inhibitor of neuraminidase, an enzyme crucial for influenza virus replication. By blocking this enzyme, Tamiflu effectively prevents the spread of the virus within the body.
Key Benefits of Tamiflu:
- Reduces influenza symptoms: Tamiflu significantly shortens the duration of flu symptoms by up to 32 hours.
- Alleviates disease severity: Clinical studies have shown a 38% reduction in influenza severity in patients treated with Tamiflu compared to placebo.
- Prevents influenza complications: Tamiflu can help prevent serious complications such as bronchitis, pneumonia, and sinusitis, especially in young patients and those with underlying health conditions.
- Protects against influenza infection: Tamiflu can effectively prevent influenza infection in individuals at high risk, including those in close contact with infected individuals, the elderly, and those with weakened immune systems.
Dosage and Administration:
The recommended dosage of Tamiflu varies based on the purpose (treatment or prevention) and patient’s age and weight. It is typically taken orally, with or without food, for 5 days for treatment and up to 10 days for prevention. For children under 1 year, Tamiflu capsules are not recommended.
Special Considerations:
- Kidney damage: Dose adjustments are required for patients with varying degrees of kidney impairment.
- Hepatic impairment: While dose adjustments are not generally needed for mild to moderate liver impairment, the use of Tamiflu in patients with severe hepatic impairment is not recommended.
- Elderly patients: Dose adjustments are typically not required for the elderly.
- Pregnancy and breastfeeding: Tamiflu should be used during pregnancy and breastfeeding only when the potential benefit to the mother outweighs the potential risks to the fetus or infant.
Potential Side Effects:
The most common side effects of Tamiflu include nausea, vomiting, diarrhea, and abdominal pain. In rare cases, more serious side effects such as convulsions and delirium-like neuropsychiatric disorders have been reported.
Drug Interactions:
Tamiflu is generally safe to use with other medications. However, it is important to inform your doctor about all medications and supplements you are currently taking to avoid potential interactions.
Important Precautions:
- Resistance: While Tamiflu is generally effective, resistance can occur. The seasonal sensitivity of the circulating influenza virus should be considered when using Tamiflu.
- Not a substitute for vaccination: Tamiflu is not a replacement for influenza vaccination.
- Safe disposal: Do not flush Tamiflu capsules down the toilet or dispose of them in household waste. Follow instructions provided by your pharmacist or doctor for safe disposal.
Consult a Healthcare Professional:
This information is not a substitute for medical advice. Always consult with your doctor or healthcare provider for any questions or concerns regarding Tamiflu or influenza.
Keywords:
Tamiflu, oseltamivir, influenza, flu, treatment, prevention, antiviral, capsules, dosage, side effects, interactions, neuraminidase, resistance, vaccination, safety, disposal, healthcare, doctor, medication, pharmaceutical, health, virus, infection, symptoms, complications, clinical studies, benefits, risks, precautions, guidelines, information, guide, comprehensive, detailed, easy to understand, SEO optimized.
| INN | oseltamivir |
|---|---|
| The code | 97 976 |
| Barcode | 300 040 800 852 |
| Dosage | 75mg |
| Active substance | Oseltamivir |
| Manufacturer | Delpharm Milano S.R.L., Italy/Hoffmann-La Roche, Switzerland |
| Importer | Foreign trade unitary enterprise "AVAFARMA", 220020, Minsk, Pobediteley Ave., 103, office 1 (7th floor); SZAO "Medvaks", Minsk, Republic of Belarus, 220002, Minsk, st. V. Khoruzhey, 31 letter A 1/K, VSTR, 1st floor. |
Related products
Antivirals
Antivirals
 Free worldwide shipping on orders $99+
Free worldwide shipping on orders $99+  US: temporary delays — postal services aligning new import rules,
US: temporary delays — postal services aligning new import rules,  EU: 1–2 weeks,
EU: 1–2 weeks,  Worldwide: 1–4 weeks
Worldwide: 1–4 weeks 

 Olive Green Convertible Multi-Purpose Tote Bag SURT-01
Olive Green Convertible Multi-Purpose Tote Bag SURT-01  Grindex Mildronate, 500 mg, 60 caps.
Grindex Mildronate, 500 mg, 60 caps.