-
×
 Pastilles Naturino Raspberry with vitamins and natural juice №8
1 × $9.00
Pastilles Naturino Raspberry with vitamins and natural juice №8
1 × $9.00 -
×
 Orthopedic insoles for adults art. D2-2 p.45 diabetic unloading.
1 × $19.00
Orthopedic insoles for adults art. D2-2 p.45 diabetic unloading.
1 × $19.00 -
×
 210cm Semi-Folding Ramps (Modular, Right-Side Mount)
1 × $279.00
210cm Semi-Folding Ramps (Modular, Right-Side Mount)
1 × $279.00 -
×
 Foot Corrector 1227
1 × $399.00
Foot Corrector 1227
1 × $399.00 -
×
 Women's Prescription Eyeglass Frames
1 × $19.00
Women's Prescription Eyeglass Frames
1 × $19.00 -
×
 Gynecological set Juno type 4M+
1 × $9.00
Gynecological set Juno type 4M+
1 × $9.00 -
×
 Belt medical elastic size 4
1 × $19.00
Belt medical elastic size 4
1 × $19.00 -
×
 UltraBalance Immune Support: Omega-3 & Vitamin D3 (90+180 Capsules)
1 × $119.00
UltraBalance Immune Support: Omega-3 & Vitamin D3 (90+180 Capsules)
1 × $119.00 -
×
 D'Arco Method
1 × $129.00
D'Arco Method
1 × $129.00 -
×
 Herbion Linkas with orange flavor, 16 pastes.
1 × $19.00
Herbion Linkas with orange flavor, 16 pastes.
1 × $19.00 -
×
 Queen Pillow, 60x60cm: Luxurious Comfort
2 × $99.00
Queen Pillow, 60x60cm: Luxurious Comfort
2 × $99.00 -
×
 Ecocaps Air & Surface Disinfectant - 10 Capsules
1 × $19.00
Ecocaps Air & Surface Disinfectant - 10 Capsules
1 × $19.00 -
×
 Orthopedic Pillow 50x70cm, 12cm Height: Memory Foam Comfort
1 × $69.00
Orthopedic Pillow 50x70cm, 12cm Height: Memory Foam Comfort
1 × $69.00 -
×
 Napkins sterile 8-layer size 50mm x 50mm №3
1 × $9.00
Napkins sterile 8-layer size 50mm x 50mm №3
1 × $9.00 -
×
 Multivitamins for Women & Men - 120 Tablets
1 × $59.00
Multivitamins for Women & Men - 120 Tablets
1 × $59.00 -
×
 Syringe two-component BD Discardit II 5 ml with a needle 22G x 1 1/2
1 × $9.00
Syringe two-component BD Discardit II 5 ml with a needle 22G x 1 1/2
1 × $9.00 -
×
 Olive Green Convertible Multi-Purpose Tote Bag SURT-01
2 × $109.00
Olive Green Convertible Multi-Purpose Tote Bag SURT-01
2 × $109.00 -
×
 Best Toe Separators
1 × $939.00
Best Toe Separators
1 × $939.00 -
×
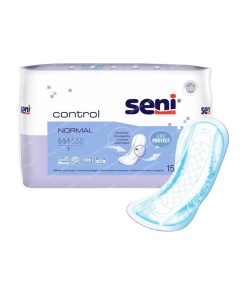 SENI CONTROL Urological pads in size NORMAL 15 pcs
1 × $19.00
SENI CONTROL Urological pads in size NORMAL 15 pcs
1 × $19.00 -
×
 Sensory Room Equipment: Balance Board & Tactile Panel for Kids
1 × $59.00
Sensory Room Equipment: Balance Board & Tactile Panel for Kids
1 × $59.00 -
×
 Pine Extract Briquettes (Direct from Manufacturer)
1 × $19.00
Pine Extract Briquettes (Direct from Manufacturer)
1 × $19.00 -
×
 55mm Ashless Filter Papers, White Ribbon, 100 Count
2 × $9.00
55mm Ashless Filter Papers, White Ribbon, 100 Count
2 × $9.00 -
×
 Rubicon Venolex, 450 mg, 60 tablets
1 × $42.50
Rubicon Venolex, 450 mg, 60 tablets
1 × $42.50 -
×
 Back, Neck & Shoulder Massager: 6-Roller, Spiked & Anti-Hunching Device + Sadhu Board
1 × $399.00
Back, Neck & Shoulder Massager: 6-Roller, Spiked & Anti-Hunching Device + Sadhu Board
1 × $399.00 -
×
 Hellebore Root, 250g
1 × $39.00
Hellebore Root, 250g
1 × $39.00 -
×
 Syringe Avanti Medical three-component single use sterile with a needle 5.0 ml
1 × $9.00
Syringe Avanti Medical three-component single use sterile with a needle 5.0 ml
1 × $9.00 -
×
 Anal Sex & Fisting Lubricant - Cool Citrus 500ml
1 × $19.00
Anal Sex & Fisting Lubricant - Cool Citrus 500ml
1 × $19.00 -
×
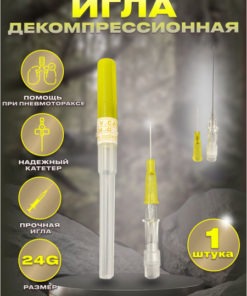 24G Blue Butterfly Catheter with Decompression Needle (Pneumothorax Treatment)
1 × $19.00
24G Blue Butterfly Catheter with Decompression Needle (Pneumothorax Treatment)
1 × $19.00 -
×
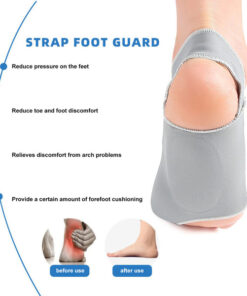 Bursitis Joint Corrector
1 × $389.00
Bursitis Joint Corrector
1 × $389.00 -
×
 CE6-35 BK Protective Caps (50 Pack)
1 × $39.00
CE6-35 BK Protective Caps (50 Pack)
1 × $39.00 -
×
![Smith Frequency 086: [Relevant Keyword] Guide](https://globalhealingweb.com/wp-content/uploads/2024/11/6295017024-247x296.jpg) Smith Frequency 086: [Relevant Keyword] Guide
1 × $139.00
Smith Frequency 086: [Relevant Keyword] Guide
1 × $139.00 -
×
 CHARM 647 C1 Eyeglass Frames
1 × $39.00
CHARM 647 C1 Eyeglass Frames
1 × $39.00 -
×
 Orthopedic insoles for men (UE, ST-105.1/ST-105.B, r40)
1 × $39.00
Orthopedic insoles for men (UE, ST-105.1/ST-105.B, r40)
1 × $39.00 -
×
 Pastilles Naturino Strawberry with vitamins and natural juice №8
1 × $9.00
Pastilles Naturino Strawberry with vitamins and natural juice №8
1 × $9.00 -
×
 VladMiVa Podolgest Titanium Thread 14 (0.35mm) - 10 Pieces
1 × $79.00
VladMiVa Podolgest Titanium Thread 14 (0.35mm) - 10 Pieces
1 × $79.00 -
×
 Copper Compression Ankle Support (2 Pack) - Injury & Sprain Prevention
1 × $19.00
Copper Compression Ankle Support (2 Pack) - Injury & Sprain Prevention
1 × $19.00 -
×
 Elbow Ice Pack
1 × $409.00
Elbow Ice Pack
1 × $409.00
Subtotal: $4,343.50
 Free worldwide shipping on orders $99+
Free worldwide shipping on orders $99+
 US: 2–3 weeks,
US: 2–3 weeks,
 EU: 1–2 weeks,
EU: 1–2 weeks,
 Worldwide: 1–4 weeks
Worldwide: 1–4 weeks